The Medicinal Chemistry Research Group of the Research Centre for Natural Sciences of the Hungarian Academy of Sciences (HAS) participates in a research programme led by Richard Moriggl from the University of Veterinary Medicine and the Ludwig Boltzmann Institute for Cancer Research, in collaboration with the group of Patrick Gunning at the University of Toronto, which aims to renew the therapeutic options for the treatment of AML, the most common type of aggressive leukemia in adults affecting the blood-forming cells originating from the bone marrow.
The examination of the development of hematogenous tumours produced a very important result in 2007: a mutant variant of a protein (codenamed JAK2) was observed in a large part of hematogenous tumours. This made it possible to identify cancer with a quick blood test in over 90% of the cases, before the occurrence of the actual symptoms of the disease.
The mutant protein was in the focus of the research of American scientist Peter Sayeski when he asked György Miklós Keserű’s team to help develop an effective inhibitor with the procedure developed by them. The procedure makes use of the unique grooves on the surface of individual proteins which enable attachment to other molecules. “We have developed methods to find these grooves and we try to tell what molecule characteristics are necessary to tightly fit the grooves,” says György Miklós Keserű. The most important aim was to find a molecule which strongly attaches to the given protein molecule preferably in a way that it cannot attach to, and thus inhibit, other (non mutant) protein molecules.
52 out of 300,000
This was the time when cooperation started with Peter Sayeski who asked the Hungarian researchers to model the structure of the newly discovered, important protein and try to find a molecule that can strongly attach to it. They looked through all 300,000 molecules in the American database and found 100 potential candidates, out of which 52 was subjected to experimental tests. Finally, of this 52 they found one about which the team could demonstrate for the first time that it:
- effectively blocks the function of the mutant protein;
- does not block the function of healthy protein;
- blocks cell division and the development of the disease in the case of tumours originating from leukemic or other hematopoietic cell lines;
- is effective in animal testing;
- is effective on human bone marrow samples.
“As this was the first molecule to block the function of only the oncogene (mutant molecule), it became an amazingly useful research tool,” György Miklós Keserű says. Although this specific molecule was not used for medication, it gave a very important tool to scientists for further research.
During a project in 2015, through the mediation of Sayeski, Richard Moriggl from the Ludwig Boltzmann Institute for Cancer Research in Austria, get into contact with the team lead by György Miklós Keserű and Dávid Bajusz. Moriggl’s team basically dealt with a different protein, called STAT5, but the two types of protein together compose a so-called intracellular signalling pathway: the upper part of which is JAK2 addressed by the Hungarian team, while the lower part was examined by the Austrian team. They noticed that the previously discovered mutation of JAK2 substantially increases the activity of the protein on STAT5 proteins, so it is a critical path to AML development. AML accounts for one-third of adult leukemia cases and has the worst outlook: the 5-year survival rate is only 26%. Due to the promising research plans, the research, which received over HUF 40 million (EUR 130000) in funding from in the discovery research funding programme of the National Research, Development and Innovation Office, has been extended to the STAT5 protein in the past three years.
Special, yet may be good for many things
In February 2018, in a scientific article published in Leukemia, a medical journal published by Nature Publishing Group, the Austrian-Hungarian-Canadian research group reports the development of a new, effective STAT5 inhibitor which successfully blocks cell division and growth in AML cell lines as well as animal models. “Our task was particularly difficult as there are very few grooves and depressions on the STAT5 protein, so it was very challenging to find a molecule that can strongly stick to it.” An important advantage of the recently developed inhibitor is that it is specific, so it will not hurt other important STAT and other proteins, but kill only the tumour cells.
Efficacy was also confirmed on human cells taken from AML patients, which not only opens up new therapeutic options in the treatment of leukemia but – according to the hopes of the consortium – in all types of cancer where STAT signalling has an essential role. The clinical development of the compound to be started in the near future and the continued research in Austria, Hungary and Canada are expected to identify further cancer diseases where the new discovery can offer new therapeutic perspectives.
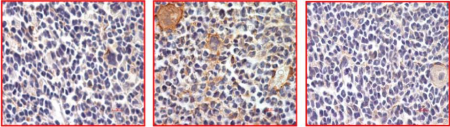 |
| STAT5 inhibition in the bone marrow |






